TECHNOLOGY SUMMARY of Cefdinir
1. Product Introduction
Cefdinir (marketed by Abbott Laboratories under the brand name Omnicef) is a semi-synthetic, broad-spectrum antibiotic in the third generation of the cephalosporin class, proven effective for common bacterial infections of the ear, sinus, throat, and skin. It was approved by the U.S. Food and Drug Administration (FDA) in December 1997.
As of 2008, cefdinir was the highest-selling cephalosporin antibiotic in the United States, with more than US$585 million in retail sales of its generic versions alone.
Chemicalname:8-[2-(2-amino-1,3-thiazol-4-yl)-1-hydroxy-2-nitroso-ethenyl]amino-4-ethenyl-7-oxo-2-thia-6-azabicyclo[4.2.0]oct-4-ene-5-carboxylic acid
MolecurFormula:C14H13N5O5S2
Molecur weight:395.41
CAS Number:91832-40-5
2. Synthetic Route
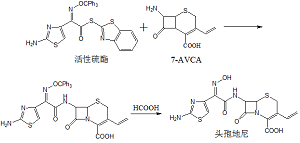
3. Product Quality
| Colour |
White Slightly yellow |
| Content |
≥ 96% |
| E-isomer |
≤ 0.5% |
| OpticalRotation |
- 58 — - 66 |
4. Contact
Add: Room 701B/D, Building B, Science Park Tower, Tsinghua Science Park, Haidian District, Beijing 100084, China
Tel: +86 10 62795180
Fax: +86 10 62795182
E-mail: biotech@ittc.com.cn
Website: www.coway.com.cn www.coway-biotech.com